Scarecrow explains
Rutherford Scattering
Between 1909 and 1911, under the direction of Ernest Rutherford, Hans Geiger and Ernest Marsden completed a series of experiments that proved that rather than being solid objects:
- Atoms contained a very small, dense nucleus.
- Most of the atom was empty space.
- All of the positive charge was found in the nucleus.
- Most of the mass of the atom was located in the nucleus.
In the experiments (see diagram), positively-charged particles – called alpha particles – which are produced by the radioactive decay of some unstable elements were fired at a very thin sheet of gold foil, which was surrounded by a circular screen, coated with a substance (phosphor) that glows when struck by the alpha particles.
The scientists found the following results:
- Most of the alpha particles hit the screen directly in front of the gold foil.
- This means that the alpha particles must have passing straight through the foil, suggesting that they weren’t hitting anything, because most of an atom is empty space.
- A few of the particles were being back-scattered, striking the screen behind the foil.
- This means that there must have been something small and dense – the nucleus – for the alpha particles to be strongly repelled from and deflected back, but it must be very small, because only a few of the alpha particles were back-scattered.
- Some of the alpha particles were striking the screen at angles to the left and right of the screen.
- This suggests that the positive alpha particles were being deflected from their normal courses by the repulsion of a like charge, telling us that the nucleus is also positive.
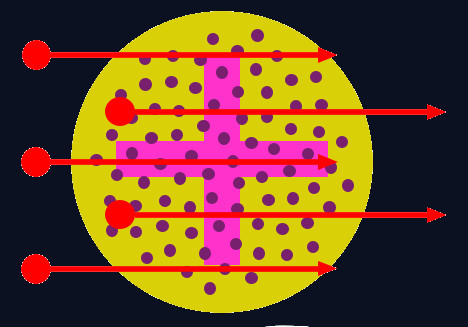
The plum-pudding model
Before Rutherford’s work, many scientists believed in J. J. Thomson’s plum-pudding model of the atom. In this model, the atom was believed to be a cloud of positive charge, with electrons evenly spread throughout it. If this was correct, alpha particles would all be expected to pass straight through the atoms with no (or at least very little) deflection or back scattering, because the positive charge would be so spread out (diffuse) that it would not be able exert a strong enough electrostatic force to repel the alpha particles and (significantly) deflect or back-scatter them.
The Dalton model
John Dalton (1766-1844) believed that atoms were small, hard, round balls (think tiny pool or snooker balls), with no structure below this, i.e. no protons, neutrons, electrons or nucleus. If John Dalton's model of atoms was correct, we would expect the alpha particles to bounce off the gold foil rather than pass through it; the fact that the alpha particles do pass through shows that John Dalton's model of atoms was wrong.
Try this in the simulation
- Click the Start source button to begin emitting alpha particles, if you have not adjusted any settings. Leave it to run for a while and then check the circular readout which shows where the particles end up: you should find about 90% of alpha particles pass straight through, with about 4.5% being scattered to the left, 4.5% to the right, and only about 1% being back-scattered.
- Click the Start source button to begin emitting alpha particles, if you have not adjusted any settings. Leave it to run for a while and then check the circular readout which shows where the particles end up: you should find about 92% of alpha particles pass straight through, with about 1% being scattered to the left, 1% to the right, and only about 6% being back-scattered.
- Note: In reality, much fewer than 1% of alpha particles would be back-scattered; it would really be about 1 in 8000 (0.0125%). The proportion of back-scattered atoms is greater than would really be the case, because the nuclei in this model, at 2% of the atom’s diameter, are far too big, they should really be 0.001% of the diameter of the atom, but that would be too small to see on the screen.
- Use the Zoom slider to zoom in on the central atom, toggle the Slow mode button on and toggle Show trace to On. Look carefully at what happens as alpha particles approach the nucleus, some strike it and are reflected back, some approach near it and are deflected from their course, most do not pass near enough to be significantly affected.
- Use the Zoom slider to zoom right out, you will see a circular Screen around the array of atoms, which flashes every time an alpha particle strikes it, over time, it will stay purple if enough atoms have struck it.
- Switch on the Plum pudding mode toggle and see how the structure of the atoms changes, and that all the alpha particles now pass straight through it, with no (significant) deflection.
- Switch on the Dalton model toggle to see what the atoms would look like in Dalton's model, and how the alpha particles just bounce off them.